87-69-4
- Product Name:L(+)-Tartaric Acid
- Molecular Formula:C4H6O6
- Purity:99%
- Molecular Weight:150.088
Product Details;
CasNo: 87-69-4
Molecular Formula: C4H6O6
Appearance: white crystals
Factory Supply High Purity High Purity L(+)-Tartaric Acid 87-69-4 Safe Transportation
- Molecular Formula:C4H6O6
- Molecular Weight:150.088
- Appearance/Colour:white crystals
- Vapor Pressure:4.93E-08mmHg at 25°C
- Melting Point:170-172 °C(lit.)
- Refractive Index:12.5 ° (C=5, H2O)
- Boiling Point:399.3 °C at 760 mmHg
- PKA:2.98, 4.34(at 25℃)
- Flash Point:209.4 °C
- PSA:115.06000
- Density:1.886 g/cm3
- LogP:-2.12260
- IDLH:621
L(+)-Tartaric acid(Cas 87-69-4) Usage
|
Chemical Properties |
white crystals |
|
Uses |
L-(+)-Tartaric Acid is a naturally occurring chemical compound found in berries, grapes and various wines. It provides antioxidant properties and contributes to the sour taste within these products. |
|
Definition |
ChEBI: A tetraric acid that is butanedioic acid substituted by hydroxy groups at positions 2 and 3. |
|
Production Methods |
Tartaric acid occurs naturally in many fruits as the free acid or in combination with calcium, magnesium, and potassium. Commercially, L-(+)-tartaric acid is manufactured from potassium tartrate (cream of tartar), a by-product of wine making. Potassium tartrate is treated with hydrochloric acid, followed by the addition of a calcium salt to produce insoluble calcium tartrate. This precipitate is then removed by filtration and reacted with 70% sulfuric acid to yield tartaric acid and calcium sulfate. |
|
General Description |
Tartaric Acid belongs to the group of carboxylic acids, and is abundantly found in grapes and wine. It is widely used in drugs, food, and beverage industry. |
|
Flammability and Explosibility |
Notclassified |
|
Pharmaceutical Applications |
Tartaric acid is used in beverages, confectionery, food products, and pharmaceutical formulations as an acidulant. It may also be used as a sequestering agent and as an antioxidant synergist. In pharmaceutical formulations, it is widely used in combination with bicarbonates, as the acid component of effervescent granules, powders, and tablets. Tartaric acid is also used to form molecular compounds (salts and cocrystals) with active pharmaceutical ingredients to improve physicochemical properties such as dissolution rate and solubility. |
|
Biochem/physiol Actions |
L-(+)-Tartaric acid serves as a donor ligand for biological processes. It is used as a food additive in candies and soft drinks to impart a sour taste. |
|
Safety Profile |
Moderately toxic by intravenous route. Mildly toxic by ingestion. Reaction with silver produces the unstable silver tartrate. When heated to decomposition it emits acrid smoke and irritating fumes. |
|
Safety |
Tartaric acid is widely used in food products and oral, topical, and parenteral pharmaceutical formulations. It is generally regarded as a nontoxic and nonirritant material; however, strong tartaric acid solutions are mildly irritant and if ingested undiluted may cause gastroenteritis. An acceptable daily intake for L-(+)-tartaric acid has not been set by the WHO, although an acceptable daily intake of up to 30 mg/kg body-weight for monosodium L-(+)-tartrate has been established. LD50 (mouse, IV): 0.49 g/kg |
|
storage |
The bulk material is stable and should be stored in a well-closed container in a cool, dry place. |
|
Incompatibilities |
Tartaric acid is incompatible with silver and reacts with metal carbonates and bicarbonates (a property exploited in effervescent preparations). |
|
Regulatory Status |
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM and IV injections; oral solutions, syrups and tablets; sublingual tablets; topical films; rectal and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients. |
|
Who Evaluation |
Evaluation year: 2017 |
InChI:InChI=1/C4H6O6/c5-1(3(7)8)2(6)4(9)10/h1-2,5-6H,(H,7,8)(H,9,10)/p-2/t1-,2+
87-69-4 Relevant articles
Efficient Catalysts for the Green Synthesis of Adipic Acid from Biomass
Deng, Weiping,Yan, Longfei,Wang, Binju,Zhang, Qihui,Song, Haiyan,Wang, Shanshan,Zhang, Qinghong,Wang, Ye
supporting information, p. 4712 - 4719 (2021/01/20)
Green synthesis of adipic acid from rene...
Electrochemical oxidation of amoxicillin on carbon nanotubes and carbon nanotube supported metal modified electrodes
Ferreira, Marta,Kuzniarska-Biernacka, Iwona,Fonseca, António M.,Neves, Isabel C.,Soares, Olívia S.G.P.,Pereira, Manuel F.R.,Figueiredo, José L.,Parpot, Pier
, p. 322 - 331 (2019/07/10)
The electrolysis of amoxicillin (AMX) wa...
A cobalt-substituted Keggin-Type polyoxometalate for catalysis of oxidative aromatic cracking reactions in water
Hong, Dachao,Kitagawa, Yasutaka,Kon, Yoshihiro,Shimoyama, Yoshihiro,Tamura, Satoru
, p. 8042 - 8048 (2020/12/28)
Efficient detoxification of harmful benz...
Bimetallic AuPt/TiO2Catalysts for Direct Oxidation of Glucose and Gluconic Acid to Tartaric Acid in the Presence of Molecular O2
Ding, Jie,Jin, Xin,Lai, Linyi,Liu, Mengyuan,Sun, Yu,Wang, Jinyao,Xia, Qi,Yan, Hao,Yang, Chaohe,Zhang, Guangyu,Zhang, Wenxiang
, p. 10932 - 10945 (2020/11/23)
Tartaric acid is an important industrial...
87-69-4 Process route
-
-
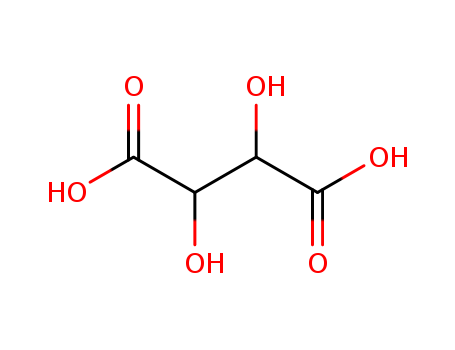
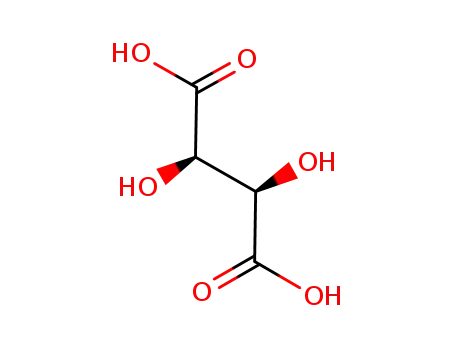
87-69-4,138508-61-9
L-Tartaric acid

-
-
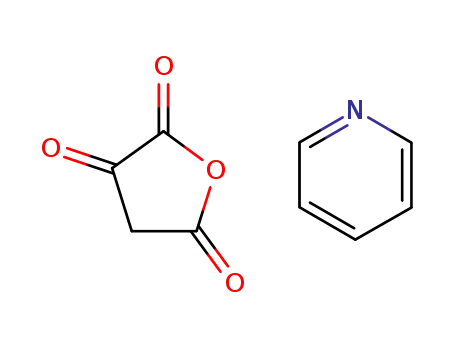
furan-2,3,5(4H)-trione pyridine (1:1)

-
-
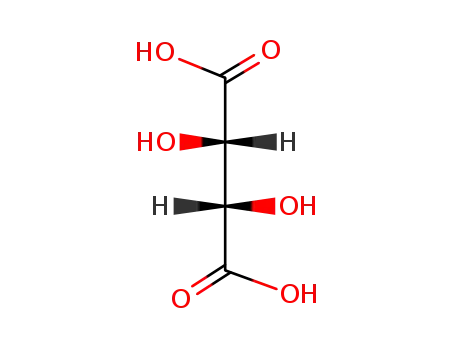
133-37-9,138508-61-9
DL-tartaric acid

-
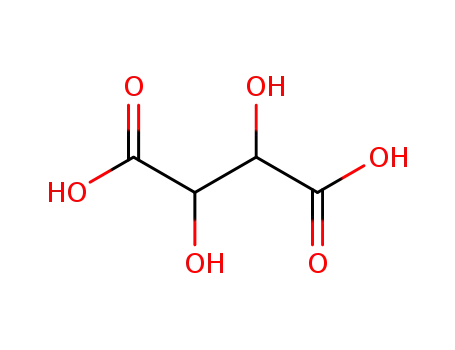
-
87-69-4,138508-61-9
tartaric acid
| Conditions | Yield |
|---|---|
|
|
-
-
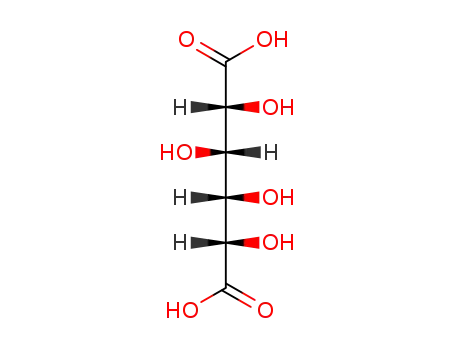
87-73-0,7231-46-1
D-glucaric acid

-

-
87-69-4,138508-61-9
L-Tartaric acid

-

-
133-37-9,138508-61-9
DL-tartaric acid
| Conditions | Yield |
|---|---|
|
With
nitric acid;
|
87-69-4 Upstream products
-
60574-00-7
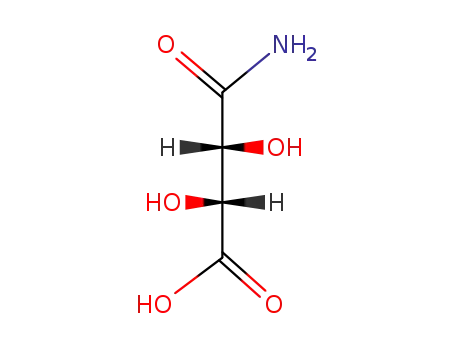
(R,R)-(+)-tartaric acid monoamide
-
87-69-4

L-Tartaric acid
-
64-17-5
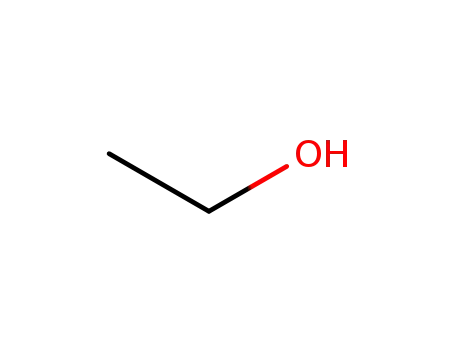
ethanol
-
902266-98-2
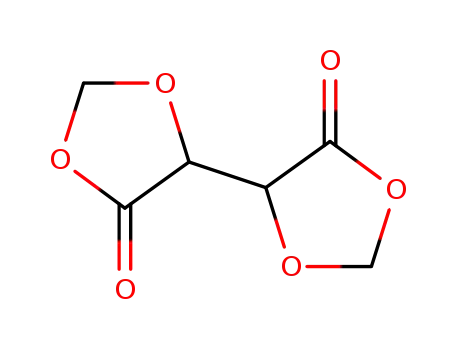
[4,4']bi[1,3]dioxolanyl-5,5'-dione
87-69-4 Downstream products
-
111955-48-7
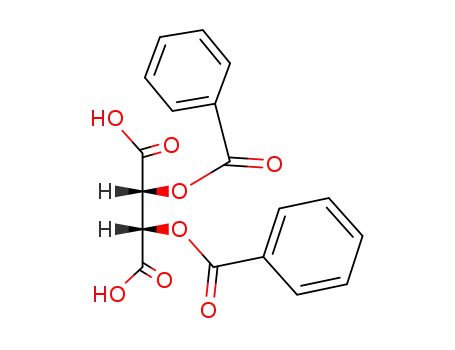
(2R,3S)-2,3-dibenzoyloxysuccinic acid
-
498-21-5
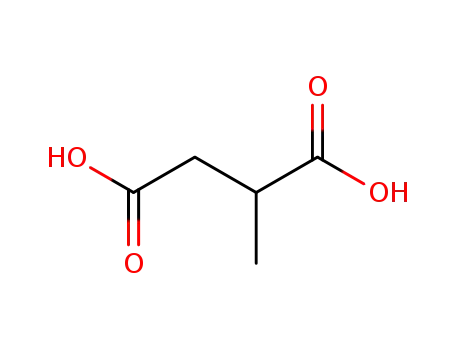
2-methylbutanedioic acid
-
51591-38-9
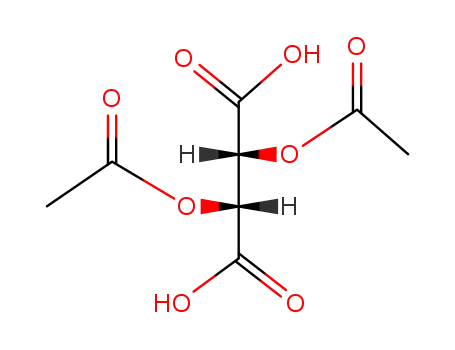
O,O′-diacetyl L-tartaric acid
-
142082-55-1
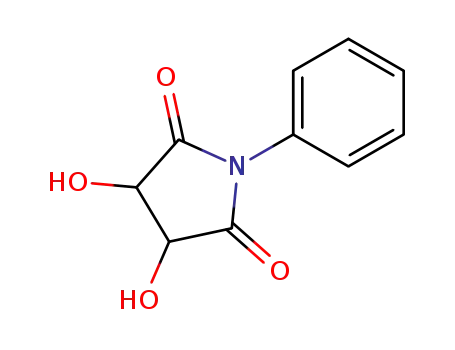
3,4-dihydroxy-1-phenyl-pyrrolidine-2,5-dione
Relevant Products
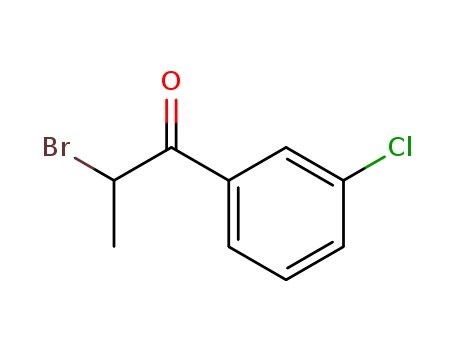
-
2-Bromo-3'-chloropropiophenone
CAS:34911-51-8
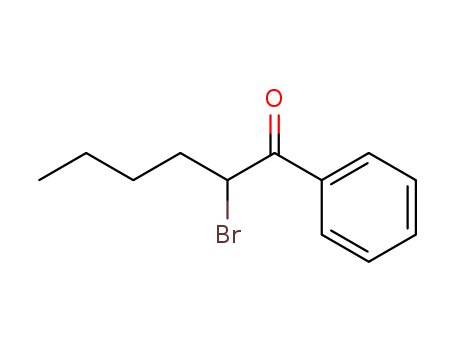
-
2-bromo-1-phenylhexan-1-one
CAS:59774-06-0
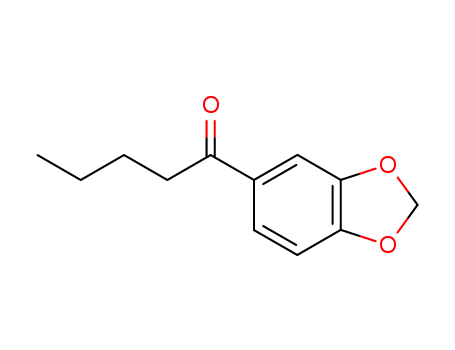
-
1-(benzo[d][1,3]dioxol-5-yl)pentan-1-one
CAS:63740-98-7