14813-01-5
- Product Name:1-Benzyl-3-piperidinol
- Molecular Formula:C12H17NO
- Purity:99%
- Molecular Weight:191.273
Product Details;
CasNo: 14813-01-5
Molecular Formula: C12H17NO
Appearance: White solid
Buy Reliable Quality High Purity 1-Benzyl-3-piperidinol 14813-01-5 Fast Delivery
- Molecular Formula:C12H17NO
- Molecular Weight:191.273
- Appearance/Colour:White solid
- Vapor Pressure:0.000631mmHg at 25°C
- Melting Point:168-172oC
- Refractive Index:n20/D 1.549
- Boiling Point:296.8 °C at 760 mmHg
- PKA:14.82±0.20(Predicted)
- Flash Point:96.3 °C
- PSA:23.47000
- Density:1.106 g/cm3
- LogP:1.58120
1-Benzyl-3-piperidinol(Cas 14813-01-5) Usage
|
Chemical Properties |
Colorless liquid |
|
Uses |
1-Benzyl-3-piperidinol is a piperidine derivative. Reactant for bioresolution of tertiary amino ester protic ionic liquids using subtilisinReactant for synthesis of:Muscarinic M3 selective antagonistsRho kinase inhibitorsPiperidine derivatives for investigations into α-adrenoreceptor direct activation |
InChI:InChI=1/C12H17NO/c14-12-7-4-8-13(10-12)9-11-5-2-1-3-6-11/h1-3,5-6,12,14H,4,7-10H2
14813-01-5 Relevant articles
N-alkylpiperidine carbamates as potential anti-Alzheimer's agents
Brazzolotto, Xavier,Gobec, Stanislav,Knez, Damijan,Kos, Janko,Nachon, Florian,?akelj, Simon,Juki?, Marko,Ko?ak, Urban,Pi?lar, Anja,Stra?ek, Nika,Zahirovi?, Abida
, (2020)
Compounds capable of interacting with si...
Transition-Metal-Free Total Synthesis and Revision of the Absolute Configuration of Pipermethystine
Quintero, Leticia,Rodr?guez-Molina, Braulio,Sartillo-Piscil, Fernando,Vázquez-Amaya, Laura Y.
, p. 3949 - 3953 (2020)
Starting from 3-hydroxy piperidines, a n...
Haemoprotein-mediated metabolism of enamines and the possible involvement of one-electron oxidations
L. M. Sayre,D. A. Engelhart,D. V. Nadkarni,M. K. M. Babu,M. E. Klein &G. McCoy
, Xenobiotica the fate of foreign compounds in biological systems Volume 25, 1995 - Issue 7
The selective prevention by cyanide of the metabolite production of 1-benzyl-3-piperidone but not 1-benzyl-3-piperidinol from 1-BP is consistent with the enamine as the source of the 3-one metabolite.
14813-01-5 Process route
-
- 64051-79-2,198976-43-1
piperidin-3-ol hydrochloride

-
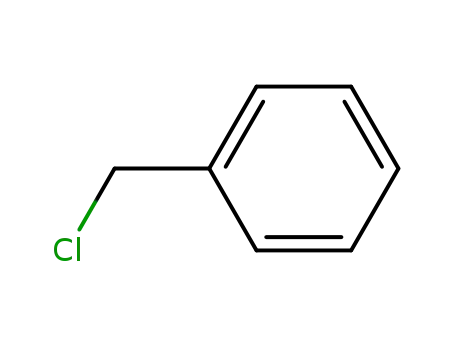
- 100-44-7
benzyl chloride

-
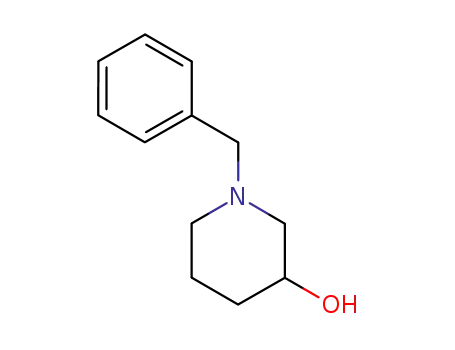
- 14813-01-5
1-benzyl-3-hydroxypiperidine
| Conditions | Yield |
|---|---|
|
With triethylamine; In toluene;
|
56.9% |
-
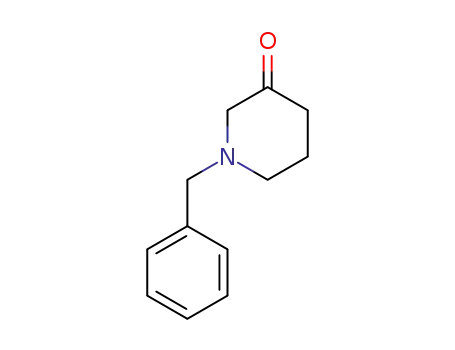
- 40114-49-6
1-benzyl-3-piperidone

-

- 14813-01-5
1-benzyl-3-hydroxypiperidine
| Conditions | Yield |
|---|---|
|
1-benzyl-3-piperidone; With sodium tetrahydroborate; In ethanol; at 20 ℃;
With hydrogenchloride; In water;
|
86% |
|
With sodium tetrahydroborate;
|
14813-01-5 Upstream products
-
3323-73-7
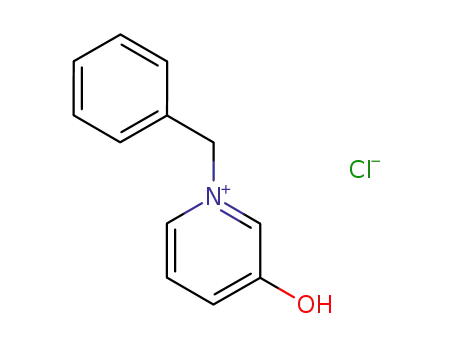
1-Benzyl-3-hydroxypyridinium chloride
-
6859-99-0
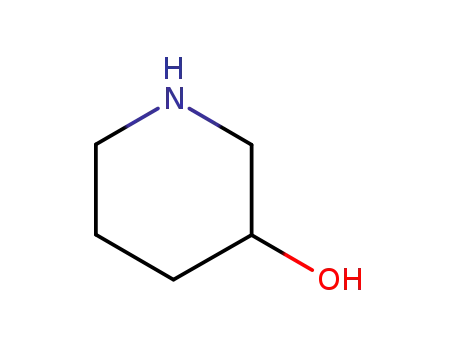
3-hydroxypiperazine
-
100-44-7
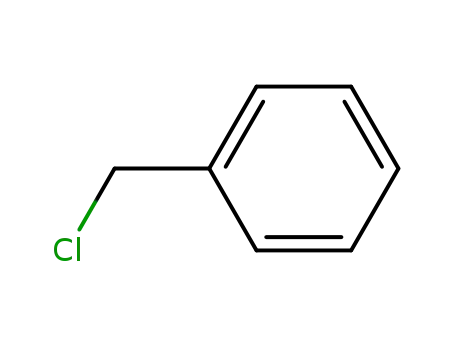
benzyl chloride
-
50606-58-1
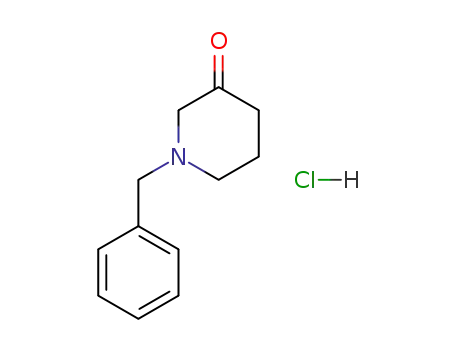
N-benzyl-3-piperidinone hydrochloride
14813-01-5 Downstream products
-
85387-34-4
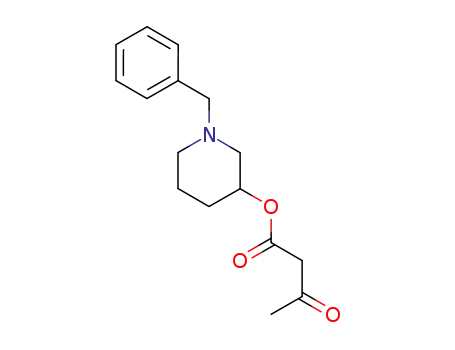
1-benzylpiperidin-3-yl acetoacetate
-
174621-91-1
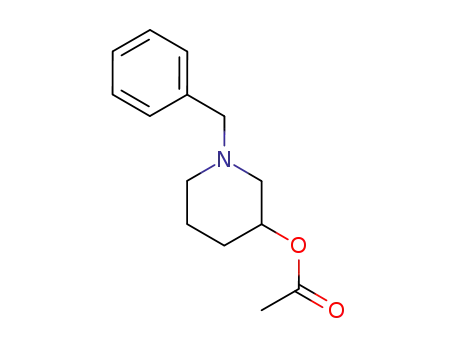
rac-N-benzyl-3-piperidinyl acetate
-
928782-46-1
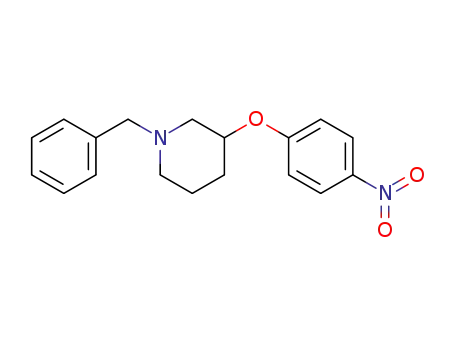
1-benzyl-3-(4-nitrophenoxy)piperidine
-
1200404-99-4
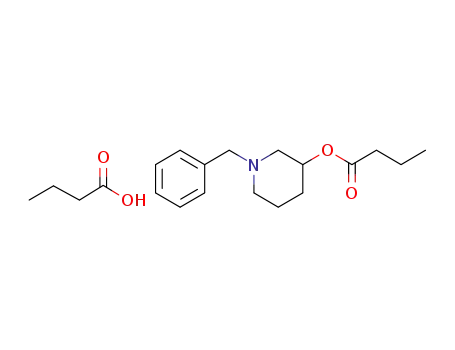
1-benzyl-3-piperidinyl butyrate butyric acid salt
Relevant Products
-
2, 5-dimethoxy-β-nitrostyrene
CAS:40276-11-7
-
Tesamorelin
CAS:901758-09-6
-
Remdesivir Intermediate
CAS:1354823-36-1